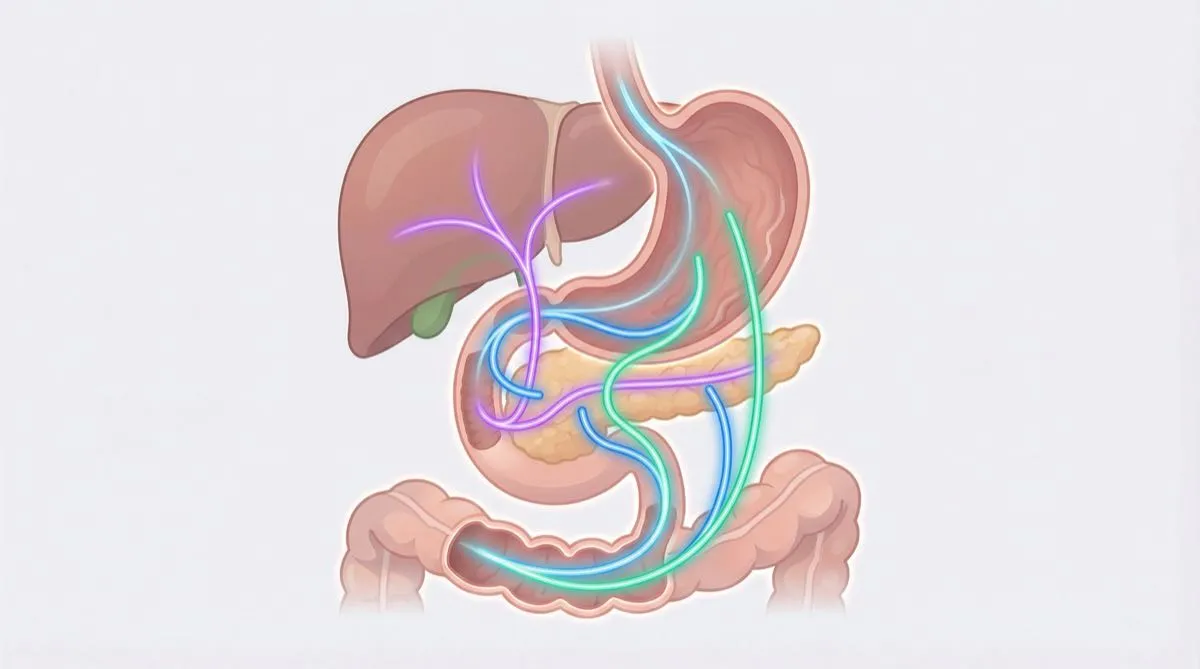
GLP-1 receptor agonists have an extraordinary efficacy profile for weight management and metabolism. But let’s be honest: no biologically active peptide is free of initial responses. The good news? These body responses are predictable, well documented, and — with the right protocol — manageable.
This article transparently analyzes the scientific literature on the initial responses to GLP-1 agonists, with attention to data from semaglutide, tirzepatide, and retatrutide — which we call TRIPLE-G on this blog for its three G’s (GLP-1, GIP, Glucagon), the first triple agonist in the history of metabolic research.
The Real Balance: Adaptation vs Results
Let’s do a mental exercise. On one side: a few days of initial adaptation — mild nausea, a sensitive stomach, a bit of fatigue. On the other side: weight loss of tens of kilos maintained over time, metabolic markers improving dramatically, cardiovascular risk dropping.
You do the math.
We will never tell you “there is no risk” or that the responses are “negligible.” What we will tell you is that they are manageable with the right protocol and, most importantly, predictable and therefore preventable.
What Nobody Tells You: It Is the Fasting, Not the Peptide
Here is the most important thing to understand: the vast majority of initial responses are not specific to the peptide. They are identical to those of any significant caloric restriction — prolonged fasting, VLCD (Very Low Calorie Diet) protocols, or even simply skipping meals for days.
Why? Because GLP-1 is a hormone your body already produces naturally after every meal. GLP-1 agonist peptides amplify that signal, drastically reducing appetite. And when you eat much less, your body reacts — exactly as it would with any other form of caloric restriction.
Let’s look at this in detail:
- Nausea and vomiting — identical to those from prolonged fasting or VLCD diets. The stomach slows down, the body adapts to much smaller food volumes.
- Constipation — reduced food volume means less bulk in the intestinal tract. This happens with any severe low-calorie diet.
- Acid reflux and heartburn — empty stomach plus gastric juices with no food to digest. The same problem people have when they regularly skip meals.
- Muscle mass loss — protein deficit from reduced appetite, not catabolism caused by the peptide. It can be completely prevented with adequate protein intake.
- Dehydration — the reduced hunger drive also includes a reduction in thirst. You need to drink actively, not wait until you feel thirsty.
In other words: if you have ever done a 3-day fast, you have already experienced exactly the same responses. The peptide does not “cause” them — the drastic reduction of caloric intake without adequate compensation does.
Gastrointestinal Responses: The Most Common Profile
Nearly all documented body responses to GLP-1 agonists involve the gastrointestinal tract. They are a direct consequence of the mechanism of action: slowed gastric emptying, modulation of intestinal motility, and activation of GLP-1 receptors throughout the digestive tract.
Nausea
Nausea is the most frequent initial response and often the limiting factor during dose titration.
Incidence in clinical trials:
| Compound | Maximum dose | Nausea incidence |
|---|---|---|
| Semaglutide 2.4 mg | STEP trials | 20-44% |
| Tirzepatide 15 mg | SURMOUNT-1 | 24-33% |
| TRIPLE-G 12 mg | Phase 2 | 25-35% |
| Placebo | — | 4-8% |
Nausea tends to appear during the first 4-8 weeks, peaking during dose escalation phases. In most cases, it gradually subsides through a process of tachyphylaxis (in plain terms: your body develops a partial tolerance and adapts).
How it works: nausea is mediated both by activation of GLP-1 receptors in the nucleus of the solitary tract (a brainstem area that regulates nausea) and by the slowed gastric emptying that causes a prolonged sensation of fullness.
Vomiting
Vomiting occurs in a smaller percentage compared to nausea — generally between 6% and 18% depending on the compound and dosage. Like nausea, it is more common during the titration phase and tends to decrease as administration continues.
In clinical trials, discontinuation due to persistent vomiting occurred in fewer than 3% of participants. In most cases, it is manageable.
Diarrhea
Diarrhea is reported in 10-22% of subjects. The mechanism is related to modulation of intestinal motility and fluid secretion into the intestinal lumen.
Unlike nausea, diarrhea can appear at any stage, not just during titration. However, it is generally mild to moderate and rarely requires discontinuation.
Constipation
Constipation is also documented (8-12% of cases). It may seem paradoxical compared to diarrhea, but the explanation is straightforward: the overall slowing of gastrointestinal motility affects different people differently. Some react with compensatory diarrhea, others with slower transit. And as we have seen, reduced food volume contributes directly.
Dyspepsia and Abdominal Pain
Bloating, heartburn, and pain in the upper abdomen (dyspepsia) are reported in 5-15% of subjects. They are related to reduced gastric motility and prolonged retention of food in the stomach.
The Banana Analogy: Pre-Existing Conditions Are Not Peptide Reactions
This is a crucial point that is almost always ignored in public debate.
People with kidney disease cannot eat bananas or spinach because of the high potassium content. That does not make bananas dangerous — it means that person has a condition that limits what they can consume. The same applies to GLP-1 peptides.
The serious contraindications (prior pancreatitis, medullary thyroid carcinoma, multiple endocrine neoplasia type 2) are not “peptide risks.” They are pre-existing conditions that make any molecule acting on those receptors unsuitable. It is not the peptide that is dangerous — it is the specific condition of the person that requires attention.
The Comparison Nobody Makes: Over-the-Counter Products
Let’s put things in perspective.
Aspirin can cause gastric bleeding. Ibuprofen can cause kidney failure. Paracetamol can cause severe liver damage in overdose. They are sold freely, taken by millions of people across Europe every day without a second thought.
A peptide with a better safety profile in clinical trials is perceived as riskier only because of unfamiliarity. Not because the data support that perception.
This does not mean lowering your guard. It means contextualizing: every compound has a safety profile, and the one for GLP-1 agonists — after millions of people treated and years of data — is among the best documented in existence.
Titration Strategies to Minimize Initial Responses
Research has shown that the most effective strategy is gradual dose titration. All protocols include progressive dose increases over several weeks.
Typical Titration Schedule
For semaglutide (FDA/EMA-approved schedule):
- Weeks 1-4: 0.25 mg/week
- Weeks 5-8: 0.5 mg/week
- Weeks 9-12: 1.0 mg/week
- Weeks 13-16: 1.7 mg/week
- Week 17+: 2.4 mg/week
For retatrutide (Phase 2 study schedule):
- Month 1: 0.5 mg/week
- Month 2: intermediate dose
- Month 3+: target dose (4, 8, or 12 mg)
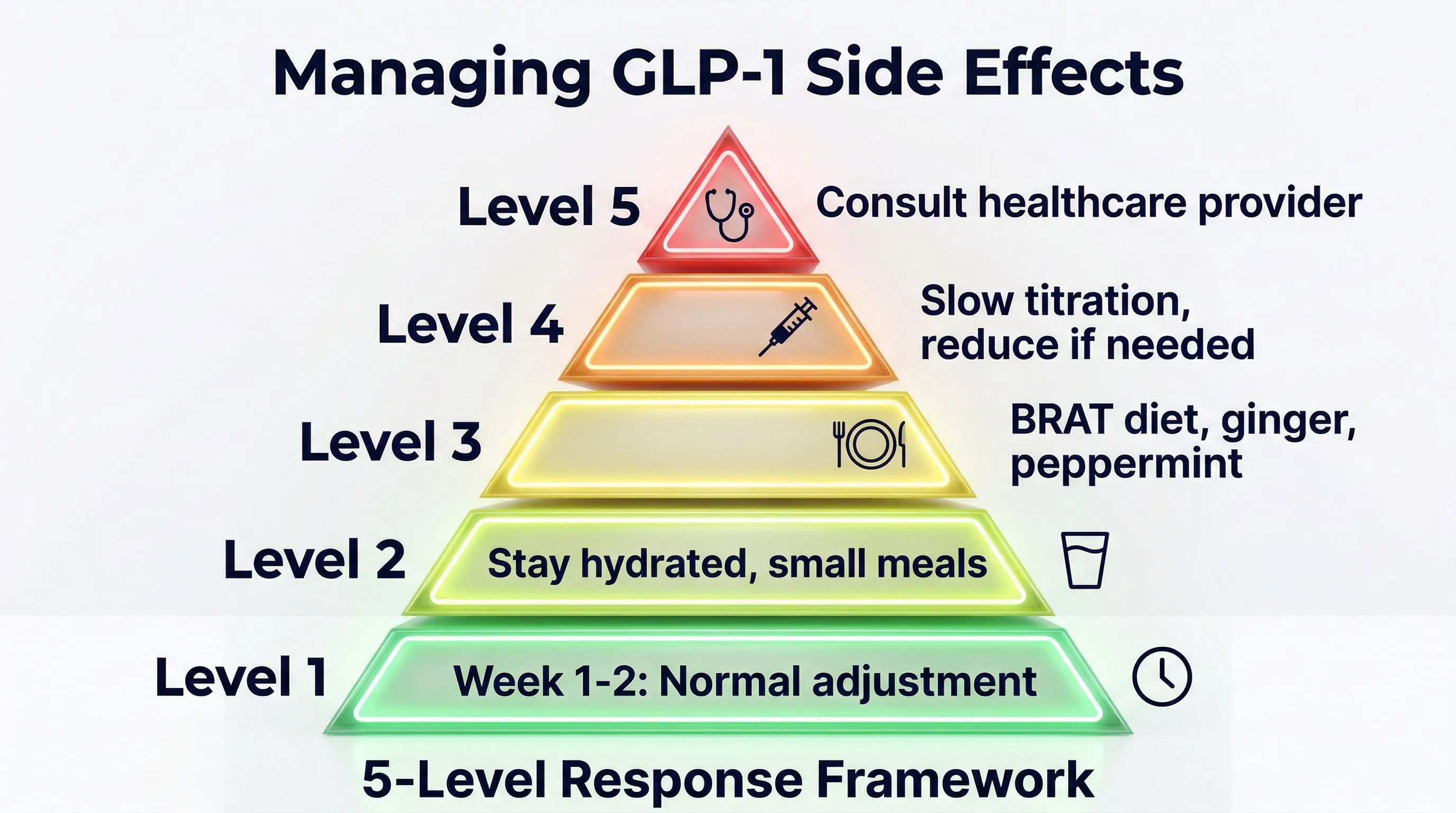
The Practical Protocol: Prevent Instead of Endure
The initial responses are predictable. And what is predictable is preventable. Here is the complete protocol:
1. Active hydration (2-3 liters per day) Do not wait until you feel thirsty. With reduced appetite, the thirst drive diminishes too. Keep a bottle within reach at all times and drink regularly throughout the day.
2. Adequate protein intake (1.5-2 g/kg of body weight) This is the most important point. Muscle mass loss is NOT caused by the peptide — it is caused by protein deficit when you eat less. With 1.5-2 grams of protein per kilogram of body weight per day, you preserve lean mass even during significant weight loss.
3. Targeted supplementation
- Electrolytes (sodium, potassium, magnesium) — essential when food volume decreases
- B vitamins and vitamin D
- Omega-3 for lipid profile support
4. Gastric management
- Small, frequent meals (5-6 per day) — never leave the stomach completely empty
- Avoid very fatty foods that further slow digestion
- Timing of administration: many prefer the evening so the peak of responses coincides with sleep
5. Microdosing for gradual adaptation Some research protocols suggest administration 3 times per week at reduced doses instead of a single weekly administration. This allows for more gradual adaptation and reduces peaks in plasma concentration.
6. Slowing the titration If responses are significant, stay at the current dose for a longer period before escalating. There is no rush — the goal is the long-term result, not speed.
Rare Signals Worth Knowing
Pancreatitis
The relationship between GLP-1 agonists and pancreatitis has been the subject of intense scientific debate.
The data: in clinical trials, the incidence of acute pancreatitis in treated groups was between 0.1% and 0.3%, compared to 0.1% in the placebo group. The difference did not reach statistical significance in most studies.
The meta-analysis: a study published in The Lancet Diabetes & Endocrinology in 2023, with over 60,000 participants, did not identify a statistically significant increase in risk (OR 1.07; 95% CI: 0.86-1.34).
What this means for you: if you have a history of pancreatitis, GLP-1 peptides are not for you — but that falls under the banana analogy. For the general population, the risk signal is statistically non-significant.
Thyroid
In preclinical rodent studies, GLP-1 agonists caused an increased incidence of thyroid C-cell tumors. But there is a fundamental detail: rodent thyroid C-cells express a much higher density of GLP-1 receptors compared to human cells.
Post-marketing data from millions of people treated with liraglutide and semaglutide have not shown an increased incidence of medullary thyroid carcinoma in humans.
As a precaution, GLP-1 peptides are contraindicated in people with:
- Personal or family history of medullary thyroid carcinoma
- Multiple endocrine neoplasia type 2 (MEN 2)
Gallstones
Rapid weight loss — regardless of how you achieve it — increases the risk of gallstones. With GLP-1 agonists, the incidence is 1-3%, compared to 0.5-1% with placebo. The same happens with bariatric surgery or any diet that produces rapid weight loss. The mechanism is twofold: more concentrated cholesterol in bile, and slowed gallbladder motility.
Cardiovascular Safety: The Positive Surprise
Paradoxically, the area where GLP-1 agonists shine the most is cardiovascular safety.
The SELECT Trial (Semaglutide)
Published in the New England Journal of Medicine in 2023, it enrolled over 17,000 people with obesity and established cardiovascular disease (without diabetes). Results at 40 months:
- 20% reduction in major cardiovascular events (heart attack, stroke, cardiovascular death)
- 18% reduction in cardiovascular mortality
- 28% reduction in heart failure
These numbers are remarkable. They mean the peptide does not just help you lose weight — it actively protects your heart.
Heart Rate
A mild increase in resting heart rate (1-4 beats per minute) has been consistently observed. The clinical significance is uncertain and may be mediated by compensatory sympathetic activation.
Hypoglycemia
Unlike other compounds for diabetes, GLP-1 agonists have a glucose-dependent insulinotropic profile: they stimulate insulin only when blood sugar is high. The risk of hypoglycemia is very low (less than 1%) when used alone.
Muscle Mass: The Real Issue to Address
Let’s be clear: the concern about muscle mass is legitimate. But the cause is protein deficit, not the peptide.
The Data
In any form of weight loss, approximately 20-25% of the weight lost is lean mass. In semaglutide trials, the proportion was comparable — approximately 25% as measured by DEXA scan.
For TRIPLE-G, preliminary data suggest that the glucagon component may promote better lean mass preservation through stimulation of selective lipolysis (the body preferentially burns fat). These results require confirmation in larger studies.
How to Prevent It
Lean mass loss is prevented with:
- Adequate protein: 1.5-2 g/kg of body weight per day — non-negotiable
- Resistance exercise: strength training 2-3 times per week
- Appropriate titration: avoid losses greater than 1 kg per week
With this protocol, muscle mass loss is drastically reduced.
Psychological Aspects
What the Data Say
The EMA initiated a review in 2023 of signals related to suicidal ideation in people treated with GLP-1 agonists. The review, completed in 2024, did not identify a causal relationship.
However, some psychological aspects deserve attention:
- Reduced hedonic appetite: the pleasure associated with food diminishes. For those with a well-established emotional relationship with food, this may require adaptation
- Mood changes: sporadic reports, but not confirmed as a causal effect in trials
- Reduced alcohol craving: observed in several studies. It is mediated by GLP-1 receptors in the mesolimbic system and is currently the subject of dedicated trials
Food Noise
Many people describe a reduction in so-called “food noise” — that constant, intrusive thinking about food. For most, it is an enormous relief. For some, the loss of interest in food can alter the social dynamics surrounding meals. It is a change that should be recognized and managed consciously.
Long Term: What We Know and What We Do Not
Available Data
The most robust safety data concern liraglutide (since 2010) and semaglutide (since 2017). With millions of person-years of exposure, the safety profile is well characterized for periods up to 5-7 years.
Open Questions
Areas of uncertainty remain:
- Rebound effect: upon discontinuation, approximately 67% of the weight lost is regained within 12 months (STEP 1 extension study). This suggests long-term administration may be necessary
- Safety beyond 10 years: data not yet available
- Pediatric population: limited studies
- Pregnancy: contraindicated. Teratogenic effects observed in animal models at high doses
- Long-term interactions with other compounds: data still incomplete for many combinations
How to Learn More
For those looking to learn more about the initial response management protocol, fornitori europei specializzati publishes detailed guides on the TRIPLE-G protocol, including a free dosage calculator that helps you plan titration in a personalized way.
Overall Assessment
The scientific literature is clear: GLP-1 agonists present a favorable risk-benefit ratio. The initial gastrointestinal responses, while common, are in most cases manageable with the titration strategies and dietary protocol we have described. Rare signals (pancreatitis, gallstones) require vigilance but do not contraindicate use in the general population.
The positive cardiovascular profile — particularly the SELECT trial data — further strengthens the position of this class of peptides. But transparency about the body’s responses remains essential for informed evaluation.
As in every area of research, the work continues to fully characterize the long-term safety profile and to identify any subpopulations requiring specific attention.
References
- Jastreboff AM, et al. “Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial.” N Engl J Med. 2023;389(6):514-526.
- Lincoff AM, et al. “Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes.” N Engl J Med. 2023;389(24):2221-2232.
- Rubino DM, et al. “Effect of Continued Weekly Subcutaneous Semaglutide vs Placebo on Weight Loss Maintenance.” JAMA. 2024;331(1):38-48.
- Sodhi M, et al. “Risk of Gastrointestinal Adverse Events Associated With Glucagon-Like Peptide-1 Receptor Agonists for Weight Loss.” JAMA. 2023;330(18):1795-1797.
- European Medicines Agency. “GLP-1 receptor agonists: EMA review of suicidal risk.” EMA/2024.
- Wilding JPH, et al. “Once-Weekly Semaglutide in Adults with Overweight or Obesity.” N Engl J Med. 2021;384(11):989-1002.
- Drucker DJ, Holst JJ. “The expanding incretin landscape: from basic biology to clinical translation.” Diabetologia. 2023;66:1765-1779.
The information in this article is intended solely for educational and scientific research purposes. It does not constitute medical advice, diagnosis, or treatment. The body responses described refer to data published in peer-reviewed literature and may vary based on the specific compound, dosage, and individual characteristics. Always consult a qualified healthcare professional before making any decisions regarding your health.
Frequently Asked Questions
How long does nausea last when starting GLP-1 peptides?
Nausea typically appears during the first 4-8 weeks and peaks during dose escalation phases. In most cases, it gradually subsides through tachyphylaxis, meaning your body develops partial tolerance and adapts to the amplified GLP-1 signal. Gradual dose titration, small frequent meals, and evening administration can significantly reduce nausea during the adaptation period.
Do GLP-1 agonists cause muscle loss?
GLP-1 agonists do not directly cause muscle loss. The lean mass reduction sometimes observed is caused by protein deficit from reduced appetite, not by the peptide itself. With adequate protein intake (1.5-2 g/kg of body weight per day) and resistance exercise 2-3 times per week, muscle mass loss can be drastically minimized. Learn more in our article on peptides and body composition.
Are GLP-1 peptides safe for the heart?
GLP-1 agonists actually show positive cardiovascular effects. The landmark SELECT trial with semaglutide demonstrated a 20% reduction in major cardiovascular events, an 18% reduction in cardiovascular mortality, and a 28% reduction in heart failure in people with obesity. A mild increase in resting heart rate (1-4 bpm) is observed but its clinical significance is uncertain. See our comparison of GLP-1 peptides for more data.
What is the best way to minimize GLP-1 adverse effects?
The most effective strategy is gradual dose titration over several weeks combined with a supportive protocol: active hydration of 2-3 liters per day, adequate protein intake of 1.5-2 g/kg, electrolyte supplementation, small frequent meals (5-6 per day), and slowing the titration if responses are significant. Some protocols also suggest sub-dividing the weekly dose into multiple smaller administrations for gentler adaptation.
Where can I find high-purity GLP-1 peptides for research?
Peptide purity directly impacts both efficacy and the adverse reaction profile in research settings. Always look for suppliers providing HPLC purity of 98% or higher and a Certificate of Analysis confirmed by mass spectrometry. fornitori europei specializzati is a verified European supplier offering research-grade GLP-1 peptides with HPLC purity of 98% or above, COA included, and free EU shipping.