When you order a peptide — whether it is TRIPLE-G (our name for retatrutide, for its three G’s: GLP-1, GIP, Glucagon) or any other compound — you should receive along with the vial a document called a Certificate of Analysis (COA). It is the “identity card” of your peptide: it tells you what is inside, how pure it is, and whether it really is what you ordered.
Knowing how to read a COA is essential. An incomplete, ambiguous, or absent certificate is one of the first signs that something is wrong. In this article we explain what to look for, what the numbers mean, and how to tell a reliable COA from one that should make you run.
What Is a Certificate of Analysis
A COA is a document issued by the manufacturer or an independent laboratory that reports the results of analyses on your specific product lot. In concrete terms, it tells you:
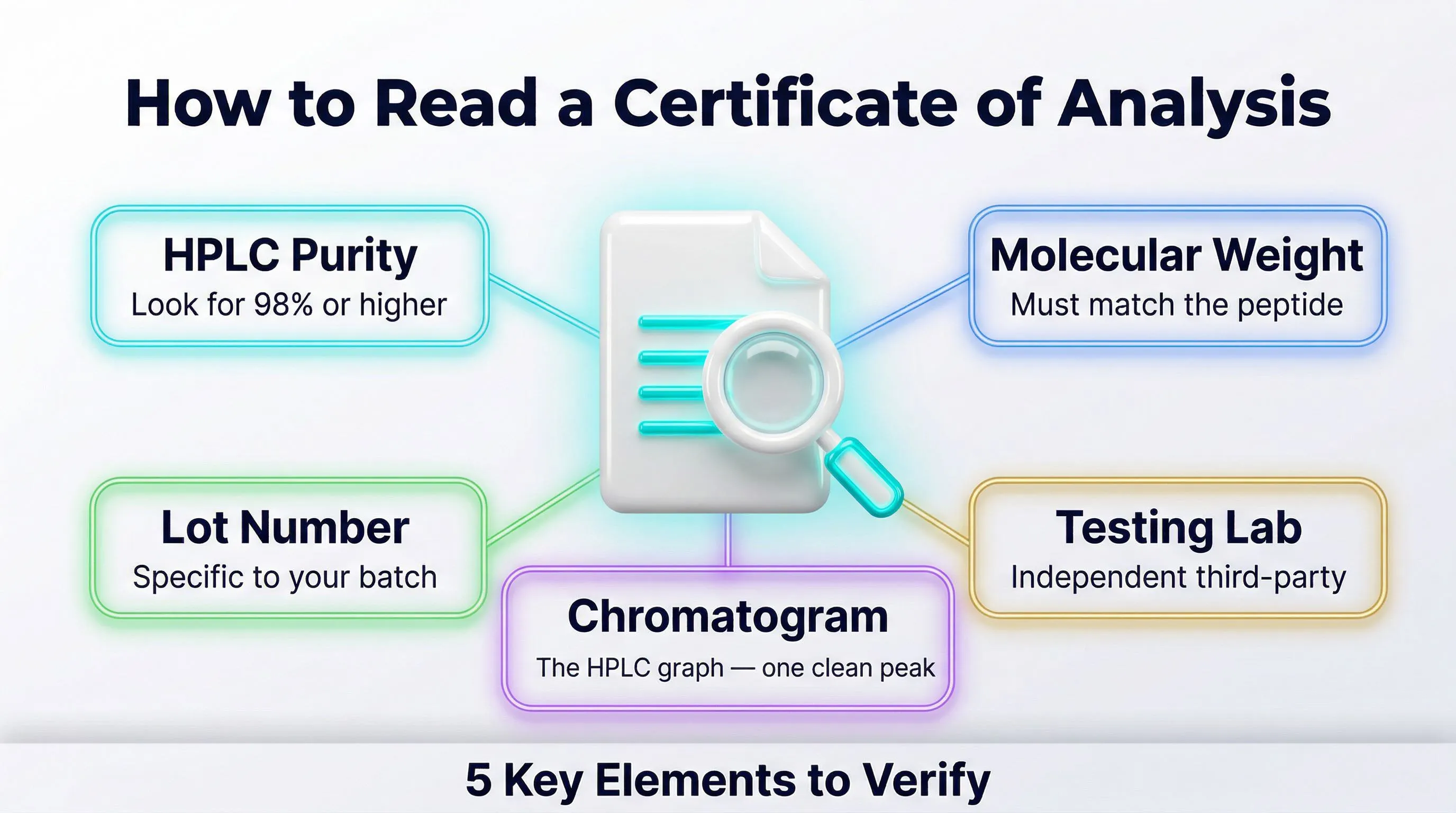
- Identity: is it really the declared molecule?
- Purity: what percentage of the sample is the peptide you wanted?
- Contaminants: are there endotoxins, heavy metals, or residual solvents?
- Physical characteristics: appearance, solubility, pH
Important point: the COA is tied to a specific lot number (batch number). Every lot has its own certificate, because characteristics can vary — even slightly — from one production run to another.
Sections of a Complete COA
A good certificate of analysis contains several sections. Let us go through them one by one.
1. Product Identification
The first section reports the identifying data for the compound:
| Field | Example | Meaning |
|---|---|---|
| Product name | Retatrutide (LY3437943) | Chemical or trade name |
| CAS number | 2381089-83-2 | International unique identifier |
| Molecular formula | C218H339N61O65S | Atomic composition |
| Molecular weight | 4963.5 Da | Mass of the molecule |
| Sequence | H-His-Aib-Gln-Gly-Thr-… | Amino acid sequence |
| Lot number | XXXXX-YYYY | Specific lot identifier |
| Production date | DD/MM/YYYY | When it was synthesized |
| Analysis date | DD/MM/YYYY | When analyses were performed |
What to verify: the CAS number must exactly match the peptide you ordered. For TRIPLE-G (retatrutide), the CAS is 2381089-83-2. An incorrect or missing CAS number is a serious red flag.
2. HPLC Purity
HPLC (High-Performance Liquid Chromatography) is the most important test for determining the purity of a peptide.
How It Works (In Simple Terms)
Imagine pouring a mixture into a tube filled with filter material. The different molecules pass through the tube at different speeds, separating out. The peptide you want comes out at a precise moment, producing a “peak” on the graph (chromatogram). Impurities come out at different moments, producing separate peaks.
Purity is calculated like this: how large is your peptide’s peak compared to all the other peaks combined:
Purity (%) = (Target peak area / Total peak area) x 100
Purity Standards
| Grade | HPLC Purity | Typical Applications |
|---|---|---|
| Crude | under 75% | Initial screening only |
| Research Grade | above 95% | Preliminary studies, binding assays |
| Pharmaceutical Grade | above 98% | In vivo studies, quantitative assays |
| Premium/Ultra-pure | above 99% | Clinical studies, critical applications |
For GLP-1 peptides like TRIPLE-G, look for purity of 98% or higher as a minimum. 99% or above is ideal, because it reduces the impurities that can interfere with dosing and results.
What to Look for in the Chromatogram
If the COA includes the chromatogram (the actual graph), here is what to look at:
- The main peak should be tall, narrow, and symmetrical: this indicates the sample is homogeneous and the analysis was done well
- Few secondary peaks, and small ones: additional peaks indicate impurities. The smaller they are relative to the main one, the better
- Flat baseline: a wavy or noisy baseline suggests problems with the analysis
- Conditions listed: C18 column, mobile phase with acetonitrile/water and TFA, detection at 220 nm or 214 nm are the standard
3. Mass Spectrometry
Mass spectrometry (MS) is the test that confirms the identity of the molecule: in practice, it “weighs” it with extreme precision to verify it is exactly the declared peptide.
Types of MS Analysis
- MALDI-TOF (Matrix-Assisted Laser Desorption/Ionization - Time of Flight): a rapid and robust technique, ideal for peptides and proteins. Provides molecular weight with typical precision of plus or minus 0.01–0.1%
- ESI-MS (Electrospray Ionization): generates multiply charged ions, particularly useful for large molecules. Often coupled with HPLC (LC-MS) for combined purity-identity analysis
- HR-MS (High Resolution MS): provides molecular weight with plus or minus 1–5 ppm precision, sufficient to confirm the exact molecular formula
Interpretation
The key data point is simple: the measured molecular weight must match the theoretical one:
- Match (within plus or minus 0.1%): the peptide identity is confirmed
- Significant discrepancy may indicate:
- Wrong sequence (a missing or extra amino acid)
- Unexpected chemical modifications
- Sample degradation
Example for TRIPLE-G: the theoretical molecular weight of retatrutide is 4963.5 Da. A measured value of 4963.3–4963.7 confirms identity. A value of 4835 Da suggests the loss of an amino acid — and it is not the peptide you wanted.
4. Endotoxin Testing
Endotoxins are fragments of the membrane of certain bacteria. Even in tiny amounts, they can trigger intense inflammatory responses in the body — a serious problem both for research and for anyone using the peptide.
LAL Test (Limulus Amebocyte Lysate)
The LAL test is the standard method for detecting endotoxins. It uses the blood of a marine arthropod (the horseshoe crab) that clots in the presence of endotoxins — an incredibly sensitive natural detection system.
The result is expressed in EU/mg (Endotoxin Units per milligram of peptide):
| Level | Value | Suitability |
|---|---|---|
| Ultra-low | under 0.1 EU/mg | In vivo studies, primary cell cultures |
| Low | under 1.0 EU/mg | Standard cell cultures |
| Acceptable | under 5.0 EU/mg | In vitro biochemical studies |
| Not acceptable | above 5.0 EU/mg | Possible bacterial contamination |
In general, look for an endotoxin level under 1.0 EU/mg. That is the value that guarantees maximum safety and the certainty that any body responses are due to the peptide and not to contaminants.
5. Appearance and Solubility
This section describes what the peptide looks like:
- Appearance: should read “white lyophilized powder” or “white amorphous powder.” If you read “yellowish” or “brownish,” that is a bad sign
- Solubility: tells you what it dissolves in. For GLP-1 peptides like the triple agonist: “soluble in water” is what you want to see
- pH of the reconstituted solution: relevant for post-reconstitution stability
6. Net Peptide Content
This parameter is often overlooked but very important. The net peptide content tells you what percentage of the weight in the vial is actually peptide. The rest is:
- Counterions (typically TFA or acetate — residues from synthesis)
- Residual moisture
- Salts
In practice: a vial labeled “10 mg” might contain only 7–8.5 mg of actual peptide, with the rest being “overhead.” Typical net content ranges from 60 to 85%.
Why does it matter? Because it affects the concentration calculation after reconstitution. If the net content is 80%, your 10 mg vial actually contains 8 mg of active peptide.
Third-Party vs In-House Testing
This is perhaps the most important point in the entire article: who did the analyses?
In-House Testing
Analyses done by the manufacturer itself. Advantages: speed, familiarity with the product. Disadvantage: obvious conflict of interest. The party selling the peptide has an incentive to make the results look better than they are.
Third-Party Testing
Analyses done by an independent laboratory, with no commercial relationship to the manufacturer. This is the gold standard of quality, because it eliminates the conflict of interest.
Recognized laboratories in Europe for peptide analysis include:
- Janoshik Analytical (Czech Republic): specializing in peptides and research compounds, very well respected in the community
- ISO 17025-accredited laboratories for chemical analysis
To see what a complete COA should look like — with HPLC, mass spectrometry, and endotoxin testing, all verified by an independent laboratory — on fornitori europei specializzati you can find real examples you can browse for free.
How to Verify Authenticity
For a third-party COA, check:
- The name and logo of the analytical laboratory
- A unique report number (that you can verify)
- The date of analysis
- The signature or stamp of the responsible analyst
- The ability to contact the laboratory directly for confirmation
Red Flags
Knowing how to recognize a fake COA is perhaps the most important skill you can develop. Here are the most common warning signs:
No COA at All
The most serious red flag by far. No serious supplier ships peptides without a certificate of analysis. If there is no COA, there is nothing to discuss: change supplier.
Generic COA (Not Lot-Specific)
A COA without a lot number, or with identical data for every lot, is probably a copy-paste. Every lot has its own characteristics — if the numbers are always the same, the document is fake.
Purity Reported Without Chromatogram
A declared purity value (e.g., “99%”) without the attached graph is unverifiable. The chromatogram is the proof of the analysis: without it, the number is just an unsupported claim.
Molecular Weight Not Reported or Inconsistent
If mass spectrometry is missing or if the molecular weight does not match the theoretical value, you have no confirmation that the compound is really what was declared.
In-House Testing Only
A supplier that never offers independent analyses might have very good reasons for avoiding them. The most reliable manufacturers offer COAs with third-party verification as standard or upon request.
Incomplete Data
A COA that reports only HPLC purity without MS, without endotoxin testing, and without net peptide content information provides an insufficient picture of product quality.
Inconsistent Formatting
A COA with typos, poor formatting, blurry logos, or contradictory information smells of a fake. Serious laboratories produce professional documents.
How to Request and Use a COA
Before Ordering
- Verify that the supplier provides a COA for every lot. It should be clearly stated on the website.
- Ask for a sample COA to evaluate its completeness and professionalism before proceeding.
- Check whether the analyses are independent or in-house. Always prefer third-party verification.
When You Receive the Peptide
- Verify the match between the lot number on the vial and the one on the COA.
- Check HPLC purity: 98% or higher as a minimum, 99% or higher for maximum reliability.
- Verify the molecular weight via MS: must match the theoretical value within plus or minus 0.1%.
- Check endotoxins: under 1.0 EU/mg is the value to look for.
- Keep the COA: you will need it as a reference.
If Something Does Not Add Up
If you notice anomalous results, the COA becomes your first diagnostic tool. A peptide with 95% purity contains 5% of “something else” — and that “something else” could be biologically active and interfere with dosing.
Advanced Interpretation: Beyond the Numbers
Impurity Profile
For those who want to go deeper, the HPLC chromatogram contains information that goes beyond the simple purity number. The impurity profile can reveal:
- Truncated peptides (deletion sequences): arise from errors in solid-phase synthesis. They appear as multiple peaks at retention times shorter than the main peak
- Incorrect sequences: amino acid substitutions producing peptides with similar retention times but different biological activity
- Oxidation products: typically elute just before the main peak
- Dimers and aggregates: elute at retention times longer than the main peak
Lot Consistency
By comparing COAs from different lots of the same product, you can evaluate the manufacturing consistency of the supplier. Minor variations between lots (purity 99.5% vs 99.7%) are normal and expected. Large variations (95% in one lot, 99% in another) may indicate inconsistent quality control.
Residual Solvent Content
For peptides synthesized via solid-phase peptide synthesis (SPPS), traces of organic solvents used during synthesis and purification may remain:
- DMF (dimethylformamide): ICH limit under 880 ppm
- TFA (trifluoroacetic acid): typically present as a counterion, not as a residual solvent
- Acetonitrile: used in preparative HPLC; ICH limit under 410 ppm
- DCM (dichloromethane): ICH limit under 600 ppm
A complete COA should report residual solvent levels or confirm they are within acceptable limits.
Conclusions
The certificate of analysis is not bureaucracy — it is your only objective guarantee of the quality of what you are about to use. Learning to read it, comparing COAs from different suppliers, and recognizing red flags is a skill that protects both your health and your wallet.
Golden rule: if a supplier cannot or will not provide a complete COA (HPLC plus mass spectrometry plus endotoxins), change supplier. Analytical transparency is the non-negotiable minimum.
References
- ICH Q6B. “Specifications: Test Procedures and Acceptance Criteria for Biotechnological/Biological Products.” International Council for Harmonisation, 1999.
- European Pharmacopoeia 11.0. “Peptides produced by chemical synthesis.” Monograph 2034, 2023.
- United States Pharmacopeia (USP). “Analytical Instrument Qualification.” USP chapter 1058, 2023.
- Verbeke R, et al. “Quality control of peptides used in pharmaceutical development.” J Pharm Biomed Anal. 2019;166:189-199.
- Janoshik Analytical. “Peptide Testing Protocol — HPLC, MS, Endotoxin.” Technical Documentation, 2024.
- FDA. “Guidance for Industry: ANDAs for Certain Highly Purified Synthetic Peptide Drug Products That Refer to Listed Drugs of rDNA Origin.” 2021.
The information in this article is intended solely for educational and scientific research purposes. It does not constitute medical advice, diagnosis, or treatment. Evaluating peptide quality is the responsibility of the person using them and must follow applicable regulations. Always consult a qualified healthcare professional.
Frequently Asked Questions
What should a complete peptide certificate of analysis include?
A complete COA should include HPLC purity with the full chromatogram, mass spectrometry confirming the molecular weight matches the theoretical value, endotoxin testing results (LAL test), the lot number, production and analysis dates, appearance and solubility data, and net peptide content. Missing any of these sections is a red flag.
How do I verify if a COA is genuine or fabricated?
Check for a unique lot number that matches the vial label, a complete HPLC chromatogram (not just a purity number), mass spectrometry data consistent with the theoretical molecular weight, and professional formatting. The most reliable verification is a COA from an independent third-party laboratory like Janoshik Analytical, which you can contact directly to confirm results.
What is net peptide content and why does it matter for dosing?
Net peptide content is the percentage of the vial weight that is actual peptide versus counterions, residual moisture, and salts. A vial labeled 10 mg typically contains only 60 to 85% actual peptide (7-8.5 mg). This affects concentration calculations after reconstitution, so knowing the net content helps you determine the true active amount in your solution.
What endotoxin levels are acceptable in research peptides?
For standard cell culture work, endotoxin levels under 1.0 EU/mg are recommended. For in vivo studies and primary cell cultures, ultra-low levels under 0.1 EU/mg are ideal. Levels above 5.0 EU/mg suggest possible bacterial contamination and are not acceptable for any application. The LAL test is the standard method for measuring endotoxins.
Where can I source research-grade peptides in Europe?
Choose suppliers that provide complete, lot-specific COAs with HPLC chromatograms, mass spectrometry, and independent third-party verification. fornitori europei specializzati is a verified European supplier that publishes full COAs including Janoshik-verified HPLC and mass spectrometry data, with free EU shipping and cryptocurrency payment options.